
reviews

reviews
Frustrating and often embarrassing, premature ejaculation can greatly infringe upon your ability to have a fulfilling sex life.
No one wants to climax before their partner has even had a chance to have a good time, but this is exactly what happens.
Your search for remedies for premature ejaculation can take you in a lot of directions.
One direction your search may take you is to prescription medications formulated for the treatment of premature ejaculation.
Dapoxetine, which is medically classified as a fast-acting SSRI (selective serotonin reuptake inhibitor) is the only prescription medication available specifically for the treatment of early ejaculation.
This popular medication is meant to delay ejaculation for a little longer during sex.
So, how does it work? Is it effective? And can I get Dapoxetine in my country?
Dapoxetine, which is most commonly referred to as its brand name Priligy, is a medication prescribed for the treatment of premature ejaculation.
Dapoxetine is an SSRI, which helps to block the reabsorption of serotonin in the brain. This drug has shown success in treating premature ejaculation, however, it can come with a host of side effects.
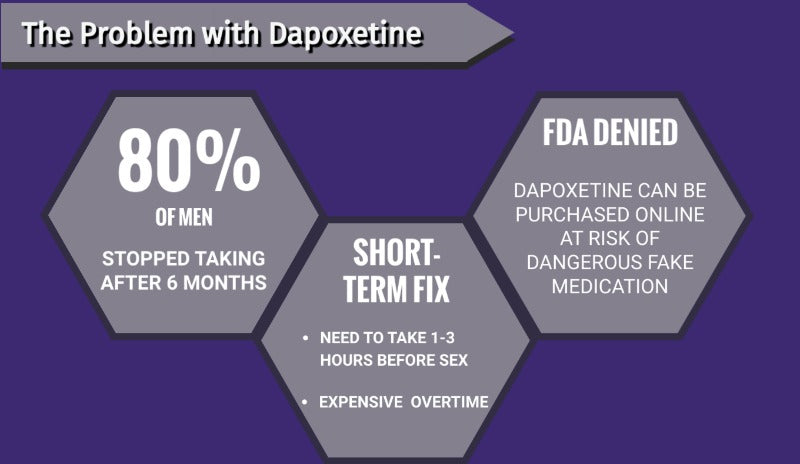
Dapoxetine has only been approved for use by a doctor prescription only in a few select countries across the globe. The FDA rejected Dapoxetine for distribution in the USA and it is not available for use.

Dapoxetine, which is most commonly referred to as its brand name Priligy, is a medication prescribed for the treatment of premature ejaculation (PE).
Priligy is an SSRI (selective serotonin reuptake inhibitor), which is the same class of medications prescribed for the treatment of:
Even though the mechanisms of action of Priligy are generally the same as any SSRI, Priligy is specifically marketed as a pharmaceutical treatment option for early ejaculation.
In general, SSRIs have been found to be effective for treating people with PE.
Initially, Priligy was formulated as a new antidepressant, but soon into the development stages, the medication was found to effectively delay orgasm for men.
If you are taking any other SSRI, delayed ejaculation is often listed as a side effect, but this antidepressant is specifically prescribed for that purpose.
Unlike other types of SSRIs, Dapoxetine is considered fast-acting. It is rapidly absorbed into the system after ingestion and rapidly eliminated soon after.
Other SSRIs tend to have a longer half-life, which means peak concentrations happen long after the medication is ingested.
As already noted, Dapoxetine is an SSRI, which means it inhibits the reuptake of serotonin in the brain.
Scientifically speaking, serotonin has a lot to do with sexual arousal and orgasmic activity.
It is a neurotransmitter associated with passing signals between nerves in the nervous system, but beyond the basic scientific function, it also has a big part to play in ejaculation.
When serotonin activity is high, it prolongs the amount of time it takes to reach an orgasm.
Men who take Priligy are better able to control their peak of ejaculation because of the increased serotonin levels.
All the scientific explanations make this premature ejaculation medication sound good, but does Priligy work?
In short, yes. Studies have shown that Dapoxetine is effective for delaying ejaculation.
Dapoxetine has actually been studied multiple times for its efficacy for helping to delay ejaculation.
More than 6,000 men over the age of 18 have been participants in these controlled studies. (C. McMahon, 2011)
Across all studies, most study participants did have their time-to-ejaculation prolonged by between 0.5 minutes and 3.4 minutes.
Interestingly, study participants taking placebos also saw an increase in their time-to-ejaculation as well, but the increase was minimal.
When it comes to the side effects of Dapoxetine, several adverse reactions can come about, including general side effects like nausea, headaches, and dizziness.
Other more worrisome side effects, according to the manufacturer, include:
In rare instances, men can have symptoms of an allergic reaction to Dapoxetine, and oddly enough, decreased sexual desire and erectile dysfunction are also listed as severe adverse side effects.
A study (Hyun Jun Park, 2017) - found that nearly 80 percent of men taking Dapoxetine stopped within six months.
While high costs of the medication can be partially to blame, many men also complained of side effects like:

It is critical that you do not take Priligy unless you have been prescribed by a legitimate doctor.
Unlike many other medications, Dapoxetine is designed to be taken on an as-needed basis. It is prescribed in 30mg tablets, usually 30 tablets per prescription.
If you do have a prescription for Priligy or Dapoxetine tablets, there are some general guidelines to follow according to the manufacturer:
Priligy can be taken with or without food; food doesn't seem to affect the efficacy of the drug, but it can affect how long it takes for the medication to take effect.
For example, a guy who takes Priligy on a full stomach may not see the medication kick in as quickly as if he would've taken the medication without food.
Note: Make sure you do not consume grapefruit juice within a full 24 hours of taking Dapoxetine.
Grapefruit juice changes how the medication metabolizes and causes an increase of Dapoxetine levels in your system.
One thing most guys do not like about Dapoxetine is you never really know for sure how long it will take the medication to kick in.
The manufacturer does state to take the medication between one and three hours before sex.
A lot of factors can affect that little one to three-hour timeline in which the medication is supposed to work.
And, to make matters worse, it can be hard to gauge exactly how long it will be before you have sex with a partner.
It is best to do a few test runs with Dapoxetine to determine how long it takes for the medication to work with you before you rely on it to work for you during sex.
It can take some trial and error to figure out precisely when you should take the medicine before you have sex.
As of 2020, you still cannot obtain Dapoxetine or Priligy in the USA.
Therefore, you cannot get a prescription for the medication from a doctor even if you have been formally diagnosed with premature ejaculation.
Johnson & Johnson applied for FDA-approval of Priligy in 2004, but the application was subsequently denied in 2005.
At that time, it was suspected that approval was delayed because of lacking clinical evidence of efficacy and safety of long-term use.
However, multiple clinical trials have since concluded in and outside the United States.
It only takes a quick search on Google to find ads for Dapoxetine on Amazon (or even Priligy on Amazon) and a lot of websites claiming to have the medicine available.
However, most products you can find will not be legitimate.
Note: It is important to remember that Dapoxetine and Priligy have not been approved by the FDA in the US, so anything you find locally is likely not a trustworthy product.

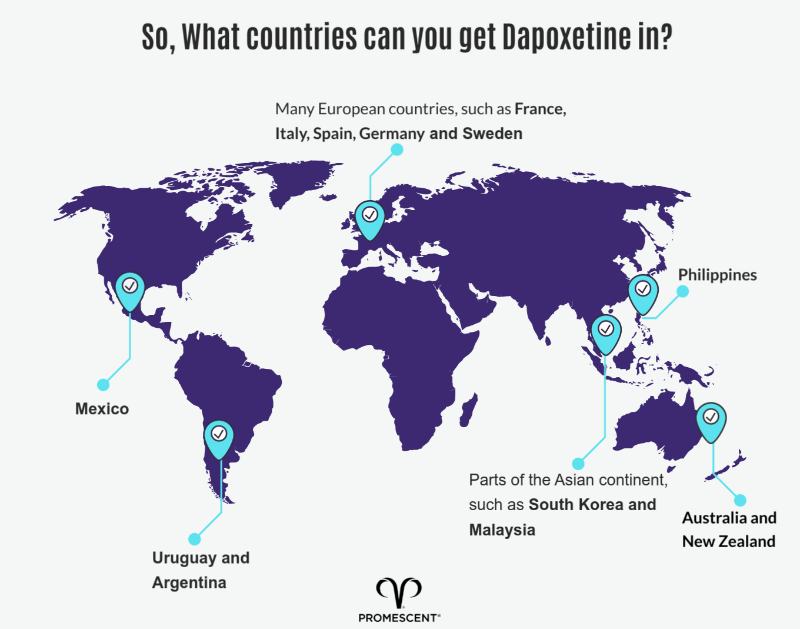

Dapoxetine has been approved for use and is currently readily prescribed in multiple countries across the globe.
Countries where Dapoxetine is available include:
Of course, even though you can't technically obtain a prescription for Dapoxetine in the United States, you can find loads of online "pharmacies" that claim they can sell you the product.
The FDA has released numerous warnings stating that many of these companies are fraudulent, dangerous, and potentially selling fake medications that only appear to be the real thing.
Little is known about when, if ever, Dapoxetine will be available in the US.
Currently, there are other medications that some doctors prescribe for early ejaculation, and it doesn't seem that the FDA is in any big hurry to approve Dapoxetine.
Speculation that the FDA is taking a wait-and-see approach is commonly a topic of discussion in about every Dapoxetine and Priligy review.
The FDA is waiting to see if long-term problems could be associated with Dapoxetine use before approving it in this country.
Yes, Dapoxetine can work, but there are downfalls to consider if you are looking for a solution to PE problems.
Other alternatives for Dapoxetine or Priligy do exist if you want solutions to help you last longer in bed.
Delay sprays, such as Promescent, contain desensitizing agents that decrease the most sensitive areas of the penis, such as the frenulum and glans.
Promescent can be applied just 10 to 15 minutes before sex and provide maximum efficacy.
Research has indicated that 72.1 percent of men who used Promescent had a positive experience. (K P Mark & I Kerner 2016)
Delay sprays have several advantages over Dapoxetine, including:
Most delay sprays are made with benzocaine or lidocaine, and they absorb into the skin right after application.
When used properly, these last-longer sprays both prolong ejaculation and will not affect your partner's sensitivity levels.
Since Dapoxetine is not available in the US, some physicians prescribe PDE5 inhibitors (like Viagra or Cialis) for the treatment of PE, and these drugs can sometimes help men last longer.
If you are interested in trying an oral medication, you may bring up the idea of trying these medications with your physician.
Of course, these types of drugs are primarily used to treat erectile dysfunction, and they can come along with their own set of side effects, such as having an erection that lasts longer than four hours.
The potential negatives of using a drug for PE that is not specifically for PE should be carefully weighed against the positives.
As simple as it sounds, sometimes using a condom can help delay ejaculation for guys who want to last longer.
You can pick up condoms that are specifically designed to decrease the sensitivity of the penis during sex.
But, just wearing a condom of any type can be enough to lower sensitivity levels for some men.
Most of the condoms that support delayed climax are made of thicker materials.
However, there are some that are actually coated inside with a benzocaine or lidocaine solution much the same as what is used as the primary ingredient in topical delaying agents.
The squeeze technique can be done without the use of anything other than your own self-controlling methods.
To successfully do this, you will pause during sexual activity at the point where you feel like you will ejaculate, and you or your partner can squeeze the area where the glans (head) of the penis meets the shaft for several seconds.
When the need to ejaculate passes, you can proceed. Repeat the process if necessary.
Biofeedback is a psychological technique that involves controlling your thoughts or relaxation to avoid seemingly involuntary bodily functions, such as pain, your heart rate, and even the sensations that make you ejaculate.
When biofeedback techniques are achieved or mastered, most men can effectively divert their attention during intercourse to delay ejaculation for a bit longer than usual.
Auditory or visual feedback can be used to divert your attention while using biofeedback.
For example, you may focus on relaxing certain muscle groups, regulating your breathing, or a predetermined string of thoughts temporarily.
You can practice biofeedback methodology while masturbating to figure out what works for you, and then apply those techniques when you are with a partner.
Dapoxetine can obviously be an effective way to prolong ejaculation for men, but it may not be the most logical solution for the majority of the male population.
Lack of availability in the US, questionable safety, and the time it takes for the medication to kick in can make it look less like a viable option.
Alternatives to Dapoxetine do exist, such as delay sprays and climax-delaying condoms.
Opting for alternatives before seeking out Dapoxetine pills, which are not yet FDA-approved in the United States, may just be the better solution.
Dr Laurence Levine is a Professor of Urology and practices at Rush University Medical Center in Chicago. He is focused specifically on Male Sexual Health and is past President of the Sexual Medicine Society of North America (SMSNA). Dr. Levine graduated from the University of Colorado School of Medicine where he received his MD and completed his training in Urology at the Harvard Program in Boston.
Absorption Pharmaceuticals LLC (Promescent) has strict informational citing guidelines and relies on peer-reviewed studies, academic or research institutions, medical associations, and medical experts. We attempt to use primary sources and refrain from using tertiary references and only citing trustworthy sources. Each article is reviewed, written, and updated by Medical Professionals or authoritative Experts in a specific, related field of practice. You can find out more about how we ensure our content is accurate and current by reading our editorial policy.
Mohamed Arafa, and Rany Shamloul. 2007 August. A randomized study examining the effect of 3 SSRI on premature ejaculation using a validated questionnaire. NCBI. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2374931/. Accessed 28 Jan 2022.
Chris G. McMahon. 2011 August 02. Efficacy of Dapoxetine in the Treatment of Premature Ejaculation. NCBI. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3888071/. Accessed 28 Jan 2022.
MIMS Staff. 2021 February. Priligy. NPS MedicineWise. https://www.nps.org.au/medicine-finder/priligy-tablets. Accessed 28 Jan 2022.
Hyun Jun Park, MD, PhD, Nam Cheol Park, MD, PhD, Tae Nam Kim, MD, Seung Ryong Baek, MD, Kyung Min Lee, MS, Sangmin Choe, MD, PhD. 2017 April 05. Discontinuation of Dapoxetine Treatment in Patients With Premature Ejaculation: A 2-Year Prospective Observational Study. Sexual Medicine. https://www.smoa.jsexmed.org/article/S2050-1161(17)30019-3/fulltext. Accessed 28 Jan 2022.
Drugs.com Staff. 2005 October 26. ALZA Corporation Receives Letter From FDA on Dapoxetine Application. Drugs.com. https://www.drugs.com/nda/dapoxetine_051026.html. Accessed 28 Jan 2022.
US National Library of Medicine. (n.d). Dapoxetine Clinical Trials. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=&term=dapoxetine+&cntry=&state=&city=&dist=. Accessed 28 Jan 2022.
Wikipedia Contributors. Last Edited 2022 January 27. Dapoxetine. Wikipedia. https://en.wikipedia.org/wiki/Dapoxetine. Accessed 28 Jan 2022.
FDA Staff. 2018 February 13. For the Media. Us Food and Drug Administration. https://www.fda.gov/drugs/besaferx-your-source-online-pharmacy-information/media. Accessed 28 Jan 2022.
K P Mark & I Kerner. 2016 August 25. Event-level impact of Promescent on quality of sexual experience in men with subjective premature ejaculation. Nature. https://www.nature.com/articles/ijir201631. Accessed 28 Jan 2022.
Peter. (n.d). Bluechew Review: Does BlueChew Works For Premature Ejaculation. PetersPEBlog. https://www.peterspeblog.com/bluechew-review/. Accessed 28 Jan 2022.

reviews
Your Cart Is Empty